|
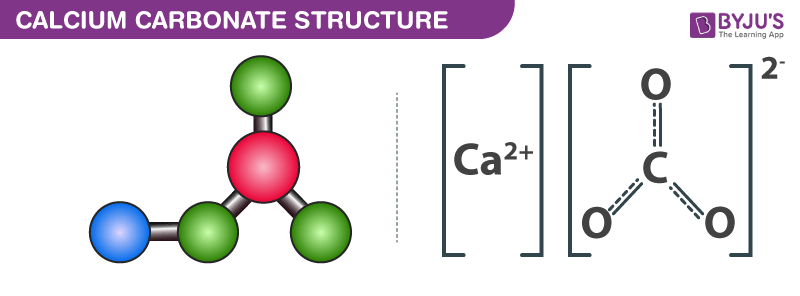
11/17/2023 0 Comments Ionic charge of calcium carbonateEven though water and CaCO 3 is ionic compound they are found to be poorly soluble in water. Let us see the solubility of CaCO 3 in water.ĬaCO 3 is not very much soluble in water. Temperature is an important factor that can alter the solubility of any solute in solvent. It has a three dimensional structure like solid with well-arranged in a regular pattern. It is a fine powder like nature with a chalky taste. Let us see whether CaCO 3 is solid or not.ĬaCO 3 is a solid which is white in colour. Solids are three dimensional shapes with height, width and length. It has triagonal planar shape with 120 0 bond angle. In this the one s and two p orbitals of carbon overlaps with p orbital of oxygen to undergo sp 2 hybridization in CaCO 3. The hybridisation is CaCO 3 Lewis structure is sp 2. The new orbitals formed as a result of hybridisation projects outwards for enhancing its overlapping ability. Calcium with two valence electrons, carbon has four and oxygen with six. The valence electrons in CaCO 3 Lewis structure is 24. Let us count the valence electrons in CaCO 3. Valence electrons has a crucial role in drawing the lewis structure of any molecule. 
So total eight lone pairs or 16 non-bonding electrons. The two oxygen atoms which single bonded with carbon has three pairs and one double bonded oxygen has two. All the lone pairs are seen in oxygen atom. 
Let us look the number of lone pairs in CaCO 3.ĬaCO 3 Lewis structure has eight lone pairs. Lone pairs usually didn’t participate in reaction but has a prominent role in shape and angle determination of a compound. So all the atoms in caco3 satisfies octet rule with eight electrons in valence shell. Calcium loses its two electrons to make its octet satisfied. Carbon and oxygen has eight electrons in their valence shell after bond making. Let us look whether CaCO 3 obeys octet or not.ĬaCO 3 lewis structure obeys octet rule. Octet rule explains about the stability of an atom after bond making. Since it has a triagonal shape the entire angle are 120 0. The bond angle of CaCO 3 lewis structure is 120 0.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
